Enter your organization-affiliated email address below to request or renew your sponsored account.
Latest Stories
Those of us who have devoted our careers to treating recalcitrant cancers know the heartbreak of walking alongside an individual facing an advanced diagnosis. We not only shoulder the clinical responsibility, but also the emotional weight that accompanies every step of that journey as each patient’s story becomes connected to our own.
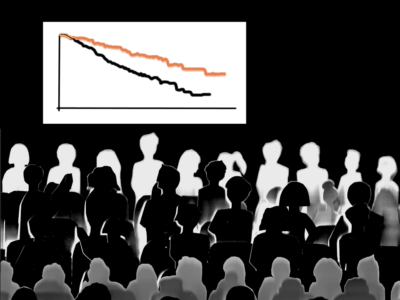
Teclistamab extends OS, deepens remissions in landmark trial published in NEJM and presented at ASCO
Houston Methodist researchers identify B cells as driver of immunotherapy resistance in liver cancer
The U.S. Department of Justice issued a letter alleging that Yale Medical School of violating anti-discrimination laws and instititing policies that disadvantaged white and Asian applicants while favoring Black and Hispanic applicants.
The DOJ investigation, which was launched a year ago, reviewed the school’s admission policies for the class of 2023, 2024, and 2025, the letter states.
“The Department finds that Yale continues to intentionally discriminate against applicants based on their race after the Supreme Court’s decision in Harvard by granting and denying admission on the basis of race,” DOJ said in the letter dated May 14.
In 2023, the U.S. Supreme Court, in its ruling of Students for Fair Admissions v. Harvard, ended the use of affirmative action.
Higher education institutions are no longer allowed to use race as a factor in their admissions processes.
“Yale has continued its race-based admissions program despite the Supreme Court and the public’s clear mandate for reform,” Harmeet K. Dhillon, assistant attorney general at the Justice Department’s Civil Rights Division, said in a statement. “This Department will continue to shed light on these illegal practices, and demand that institutions of higher education comply with federal law.”
The university issued the following statement, according to reporting from NBC Connecticut:
The students admitted to Yale School of Medicine demonstrate exceptional academic achievement and personal commitment; its program of medical education encourages curiosity and critical thinking, and its graduates go on to become leaders in clinical care, research, and public service. We will carefully review the Department of Justice’s letter. Yale School of Medicine is confident in the rigorous admissions process we follow.
This marks the second letter DOJ has sent this month to an elite medical school regarding admissions. On May 6, David Geffen School of Medicine at UCLA also received a letter with a similar allegation.
A spokesperson from UCLA noted the receipt of the letter, according to reporting by Inside Higher Ed.
We are confident in our practices and our mission to maintain access to a high-quality education to all qualified students. We are carefully reviewing the Department of Justice’s report. The David Geffen School of Medicine is committed to providing equal opportunity to all applicants and fully complying with federal and state laws.
DOJ is requesting that Yale and UCLA enter into voluntary resolution agreements to bring the schools “into legal compliance.”