Cancer Policy


Clinical Roundup


In The Clinic
Latest Issue
NEWS ALERTS
UPCOMING EVENTS
Jun 2026
Sep 2026
Conversations with The Cancer Letter
Contributors
Trending Stories
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
- Pam Bondi diagnosed with thyroid cancer
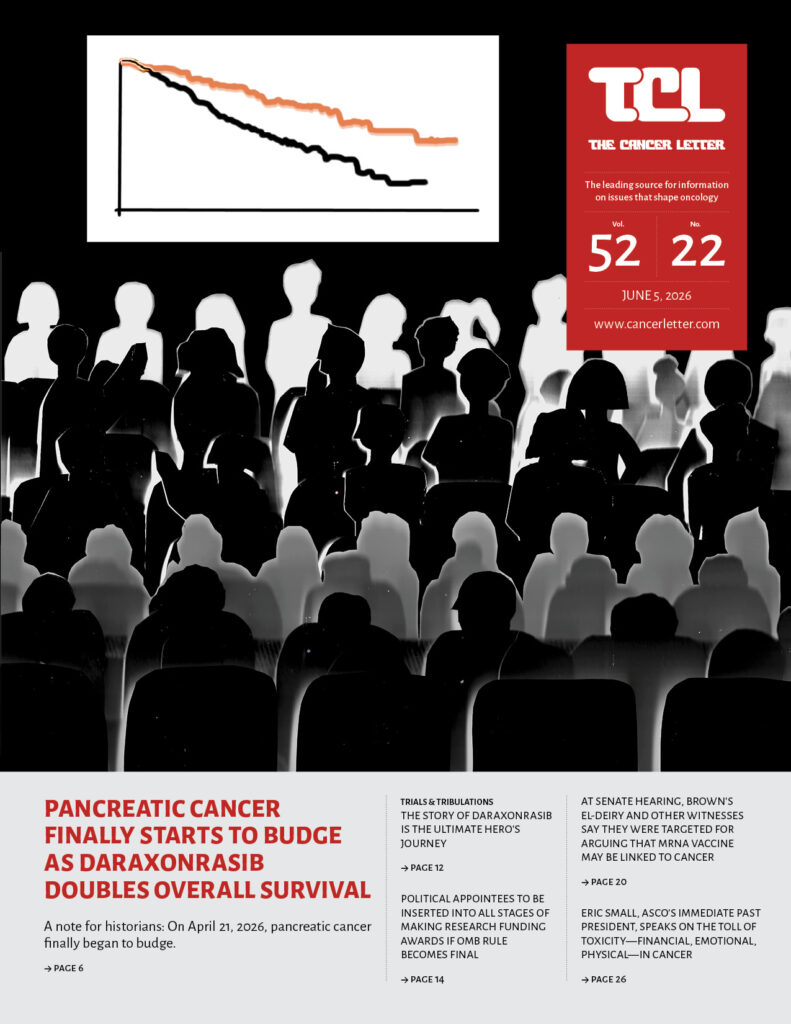
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- UK Markey’s Denise Fabian: Theranostics lets us pair a diagnostic with a radioactive therapeutic that target the same molecule
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe
Recent Issues
The Cancer Letter is an independent weekly news publication and the leading source for information on the issues that shape oncology.