Cover Story
Clinical
A story is told that after first obtaining approval for marketing Premarin in 1942, Wyeth Laboratories didn’t lock up the secret recipe for making the hormonal drug in a company safe, to be guarded by patent lawyers.
Cancer Policy
In Brief


Clinical Roundup


Drugs & Targets
Trending Stories
- Pam Bondi diagnosed with thyroid cancer
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
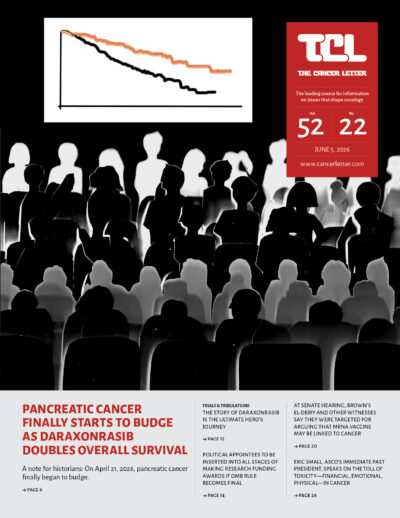
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- UK Markey’s Denise Fabian: Theranostics lets us pair a diagnostic with a radioactive therapeutic that target the same molecule
- When CAM is added to standard therapies, survival rates drop, observational study finds