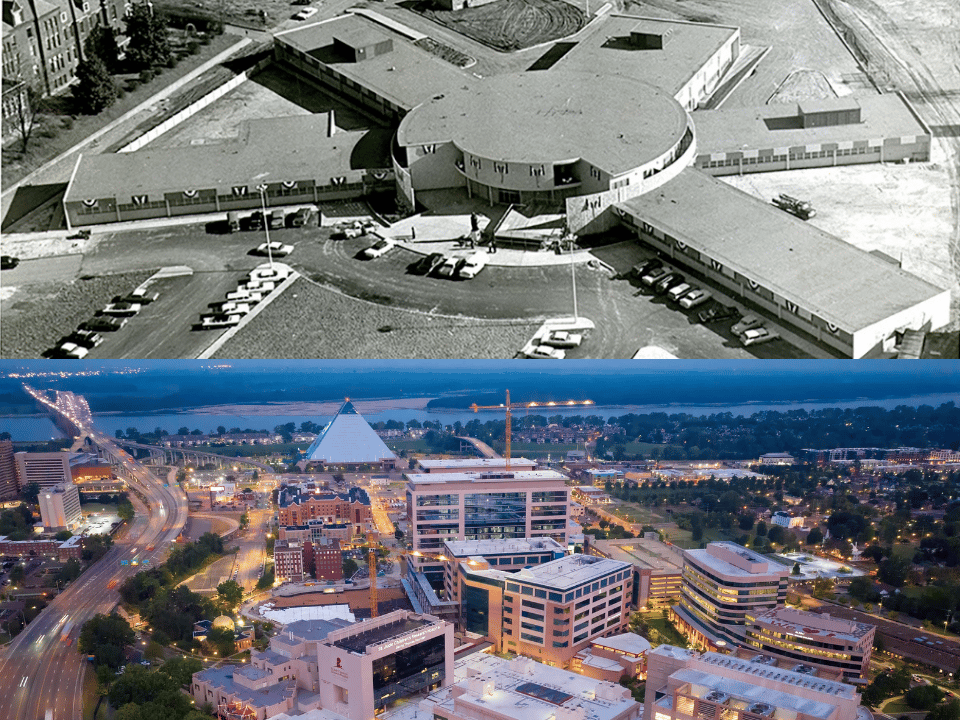Cancer Policy


Clinical Roundup


In The Clinic
Latest Issue
NEWS ALERTS
UPCOMING EVENTS
Conversations with The Cancer Letter
Contributors

Annaliese Daniels, MBA
Trending Stories
- “Backwater to Blockbuster” chronicles the previously untold story of the explosive growth of St. Jude Children’s Research Hospital
In a conversation with Deborah Doroshow, co-authors Chuck Sherr and Bill Evans discuss their collaborative writing process - Prasad’s “deceit,” plus, ODAC’s tough decision against biomarker-based progression assessment (for now)
- DOJ alleges that Yale and UCLA discriminated based on race in medical school admissions
- FDA’s second rejection of Replimune’s melanoma treatment stirs worry among oncologists, patients
“Patients are owed another review.” - Mt. Sinai forms committee to probe Epstein links to breast center founder Eva Dubin, other faculty members
- How City of Hope is turning microbiome science into better cancer care
Recent Issues
The Cancer Letter is an independent weekly news publication and the leading source for information on the issues that shape oncology.