Cover Story
Regulatory News
In a unanimous vote, the FDA Oncologic Drugs Advisory Committee advised the agency to accept the metric of “minimal residual disease,” or MRD, as a basis for accelerated approvals of therapies in all settings of multiple myeloma.
In Brief
Clinical Roundup


Drugs & Targets


Trending Stories
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
- Pam Bondi diagnosed with thyroid cancer
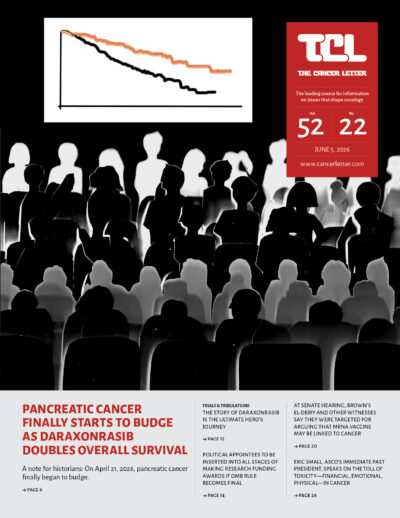
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- UK Markey’s Denise Fabian: Theranostics lets us pair a diagnostic with a radioactive therapeutic that target the same molecule
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe