Cover Story
Regulatory News
In a 14:1 vote, ODAC nixes a PD-1 drug developed in China; data not generalizable to U.S. population
By Paul Goldberg and Alice Tracey
The FDA Oncologic Drugs Advisory Committee expressed decisive support for the agency’s view that data from trials of checkpoint inhibitors performed in mainland China aren’t applicable to the U.S. population.
White House
By Matthew Bin Han Ong
In Brief


Clinical Roundup


Drugs & Targets
Trending Stories
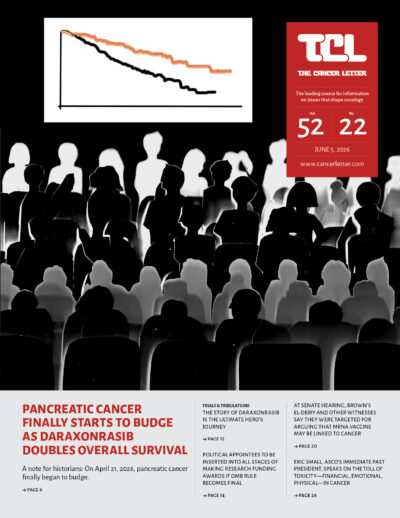
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
- Pam Bondi diagnosed with thyroid cancer
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe - As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- At Senate hearing, Brown’s El-Deiry and other witnesses say they were targeted for arguing that mRNA vaccine may be linked to cancer