Cover Story
Conversation with The Cancer Letter
By Jacquelyn Cobb, Matthew Bin Han Ong and Paul Goldberg
The past six weeks have brought fundamental change in the way oncology drugs are being developed. At this unprecedented moment in oncopolitics, FDA, NCI, academic oncologists, advocates, and the industry are in agreement on how cancer therapies should be developed, tested and approved.
In Brief


Clinical Roundup


Trending Stories
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
- Pam Bondi diagnosed with thyroid cancer
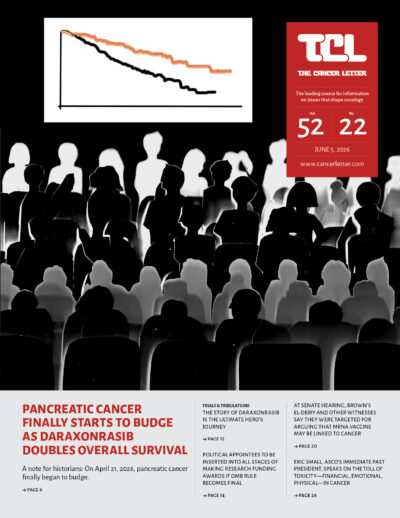
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- UK Markey’s Denise Fabian: Theranostics lets us pair a diagnostic with a radioactive therapeutic that target the same molecule
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe