Cover Story
The FDA Oncologic Drugs Advisory Committee Feb. 26 voted down a vascular-targeted photodynamic therapy for early-stage, low-risk prostate cancer and, in a narrow vote, recommended approval for a combination of a VEGF receptor 2 antagonist and an EGFR-targeting tyrosine kinase inhibitor for front-line treatment of EGFR-positive non-small cell lung cancer.
By Alexandria Carolan
In Brief
Clinical Roundup
Drugs & Targets


Trending Stories
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe - At Senate hearing, Brown’s El-Deiry and other witnesses say they were targeted for arguing that mRNA vaccine may be linked to cancer
- Pam Bondi diagnosed with thyroid cancer
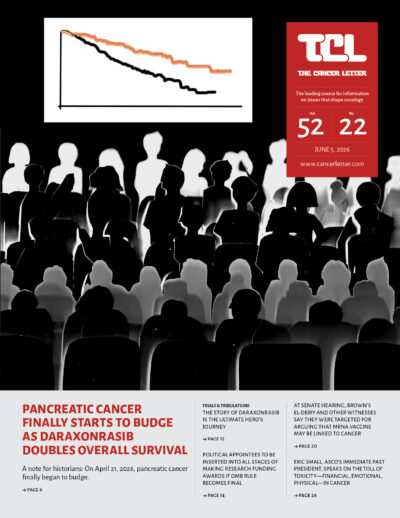
- Pancreatic cancer finally starts to budge as daraxonrasib doubles overall survival
- Wafik El-Deiry steps down as director of Brown’s Legorreta Cancer Center