
Cover Story
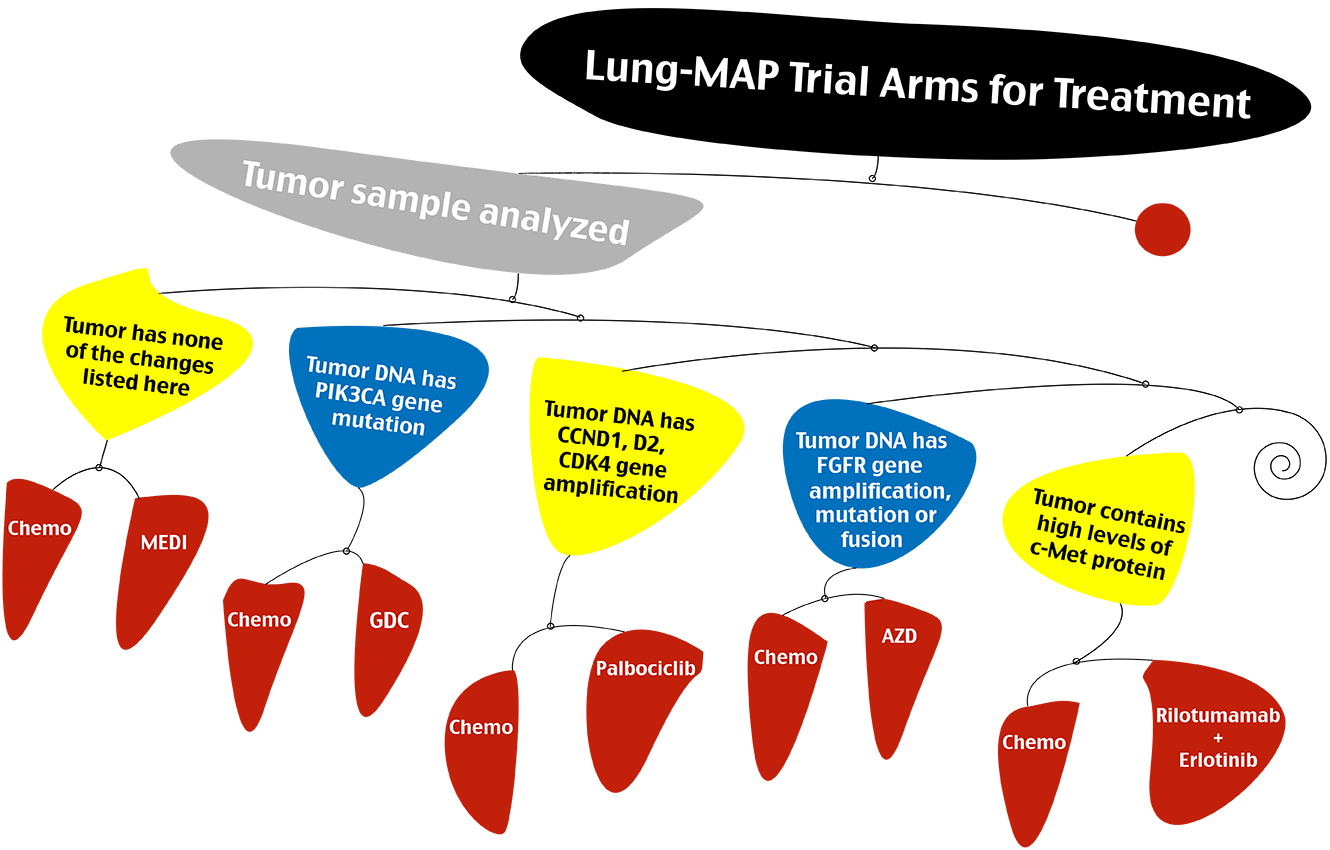
SWOG earlier this week started to accrue patients to Lung-MAP, a clinical trial for second-line treatment of non-small cell lung cancer.
By Tessa Vellek
In Brief


Trending Stories
- Kennedy calls for a “moonshot” targeted on the role of the microbiome in cancer
- Pam Bondi diagnosed with thyroid cancer
- Daraxonrasib’s success in treating pancreatic cancer is a reminder that clinical cancer researchers are heroes
- As FDA’s wheels almost entirely come off, NCI remains sturdy—a cancer moonshot may even be on the horizon
- UK Markey’s Denise Fabian: Theranostics lets us pair a diagnostic with a radioactive therapeutic that target the same molecule
- The story of daraxonrasib is the ultimate hero’s journey
A narrative curve with twists, turns, intrigue, drama—and a blow to a mighty foe